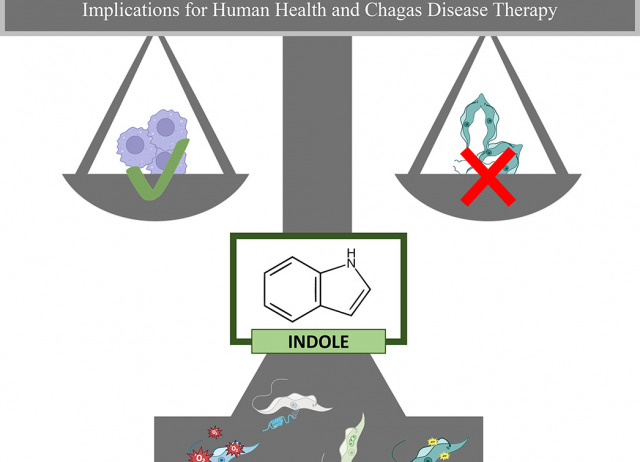
Targeting Trypanosoma cruzi with indoles: mechanistic insights and implications for human health and Chagas disease therapy
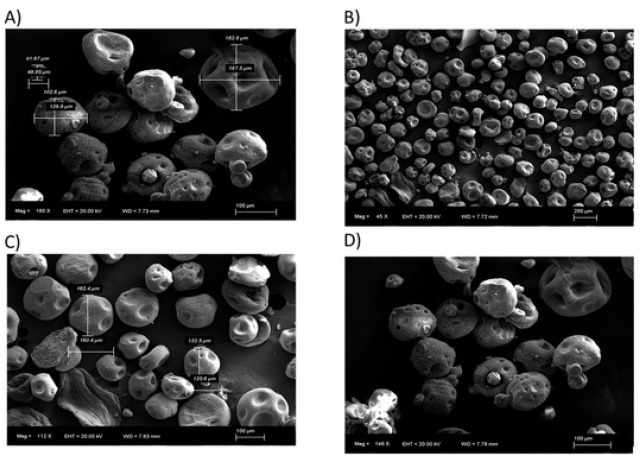
Chagas disease, caused by the protozoan Trypanosoma cruzi, remains a serious public health concern, particularly in Latin America, due to its high prevalence and the limited efficacy and safety of current treatments. To identify new potential therapeutic options, a library of 40 synthetic indole derivatives was screened for antiparasitic activity. The compounds were tested against both epimastigote and intracellular amastigote forms of T. cruzi using in vitro assays. While most of the molecules showed low or no activity, ten compounds exhibited promising effects with IC₅₀ values below 200 μM. The most active derivatives were further evaluated for cytotoxicity on mammalian cells. Among them, the compound I – 7b stood out with a selectivity index of 19.8 and was selected for further characterization. Subsequent assays revealed that this compound induced several cellular effects in the parasite, including ATP depletion, chromatin condensation, plasma membrane damage, reactive oxygen species (ROS) accumulation, and disruption of mitochondrial membrane potential (ΔΨm). These findings suggest that certain indole-based compounds may serve as promising scaffolds for the development of novel therapies against T. cruzi, contributing to the advancement of global health efforts targeting neglected tropical diseases.
C. J. Bethencourt-Estrella, A. López-Arencibia, R. Diana-Rivero, I. Sifaoui, I. M. Calero-Docina, D. Tejedor, J. Lorenzo Morales, J. E. Piñero
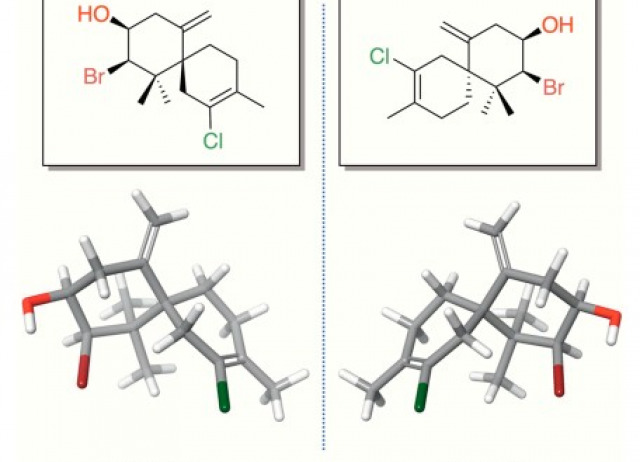
Smart Secondary Metabolites in Marine Environments: The Case of Elatol
The concept of “Smart Secondary Metabolites” is introduced here to describe a privileged class of natural products defined by structural originality, biosynthetic adaptability, and broad interaction potential with biological systems. Elatol, a halogenated sesquiterpene chiefly produced by Laurencia red seaweeds and occasionally accumulated by their consumers, exemplifies this concept with remarkable clarity. Its biosynthesis unfolds from farnesyl diphosphate via γ-bisabolane cations, bromochlorination, and stereoselective cyclization to chamigrene scaffolds, generating both (+)- and (–)-enantiomers, two metabolites with clearly distinct potential ecological roles and pharmacological profiles. This review synthesizes the current state of knowledge on elatol’s distribution, biosynthetic origins, ecological relevance, and therapeutic potential. Elatol serves as a multifunctional chemical mediator, fulfilling defensive, communicative, and regulatory roles whose intensity shifts in response to herbivory, biofouling, temperature, and salinity. In parallel, its potent activities against infectious, metabolic, and neoplastic diseases highlight its growing value as a drug lead, reflected in a rising number of patent claims. Altogether, elatol emerges as a model Smart Secondary Metabolite whose ecological sophistication and biochemical versatility position it as a promising scaffold for marine-derived drug discovery.
Soares, Angélica; Nocchi, Nathalia; Díaz-Marrero, Ana Raquel; Pereira, Renato C.; Fernández, José J.
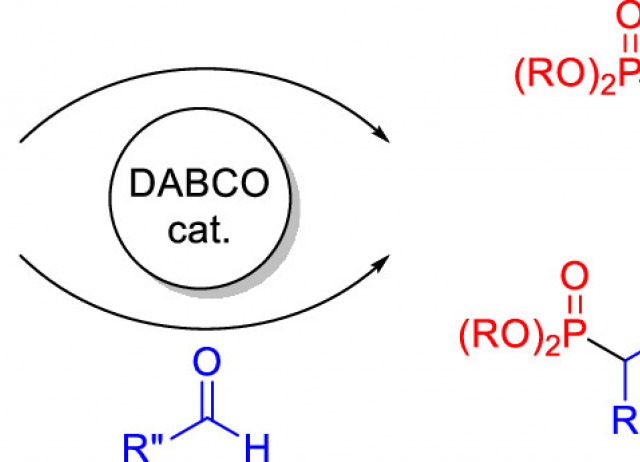
Catalytic Hydrophosphorylation of Propiolates and Three-Component Phosphorylation of Aldehydes
A practical and efficient regio- and stereoselective hydrophosphorylation of propiolates, as well as a multicomponent reaction incorporating an aldehyde component, is reported. Both processes proceed with atom economy in very straightforward experimental procedures. The reactions are catalyzed by DABCO (1,4-diazabicyclo[2.2.2]octane) and use readily available H-phosphonates as the phosphorylating agent.
S. Delgado-Hernández, A. Peixoto de Abreu Lima, E. M. Martín-Díaz, J. Scoccia, R. Carrillo, D. Tejedor
Leishmanicidal and trypanocidal potential of ß,γ-unsaturated carboxylic acid synthetic derivatives
Dedicated to the memory of Dra. Celina García, whose mentorship was essential to the development of this project. The authors express their deep gratitude to Prof. Víctor S. Martín, recently retired, for his generous contribution and financial support in the design of the synthesized products. Leishmaniasis and Chagas disease are parasitic infections with limited treatment options due to drug toxicity and rising resistance. Leishmaniasis, caused by Leishmania spp., often manifests in a cutaneous form and is treated with drugs like amphotericin B and miltefosine, which can cause severe side effects. Chagas disease, caused by Trypanosoma cruzi, is endemic in the Americas and classified by the WHO as a neglected tropical disease. Existing treatments are effective mainly in the acute phase but have significant limitations. In search of new therapies, this study explored diversity-oriented synthesis to generate a library of β,γ-unsaturated carboxylic acid derivatives. From 23 synthesized compounds −12 reported for the first time– several showed promising antiparasitic activity. Four compounds were effective against Leishmania amazonensis, with three also targeting the amastigote form. Two compounds showed activity against Trypanosoma cruzi, outperforming reference drugs in selectivity. Signs of parasite apoptosis, including mitochondrial damage and ROS accumulation, suggest strong therapeutic potential for these derivatives.
Delgado-Hernández, Samuel; Calero-Docina, Isabel María; Luis-Rancel, Sophie; Bethencourt-Estrella, Carlos J.; Álvarez-Méndez, Sergio J.; López-Arencibia, Atteneri; Lorenzo-Morales, Jacob; Piñero, José E.
Control of Tetrazine Bioorthogonal Reactivity by Rotaxanation
Rotaxanation is an efficient method to control the tetrazine-mediated inverse electron demand Diels–Alder (IEDDA) reaction. Tetrazine rotaxanes were synthesized in high yield by crown ether active template applied to the nucleophilic aromatic substitution of tetrazines. Kinetics of the bioorthogonal reaction with tetrazine rotaxanes were much slower than those with the corresponding threads. Interestingly, disassembly of the mechanical bond upon the application of the right stimulus activates IEDDA. Control of IEDDA in live cells was proved by means of a β-galactosidase sensitive tetrazine rotaxane: Enzymatic digestion allowed the Diels–Alder reaction, which resulted in a fluorescent compound.
Carlos Fumero-Medina, Yaiza Pérez-Pérez, Lidia A. Pérez-Márquez, Diego Álvarez de la Rosa, Teresa Giráldez, Nieves Rodríguez Paz, Jorge Pasán, Fernando Lahoz, David Tejedor, Jimena Scoccia, Romen Carrillo
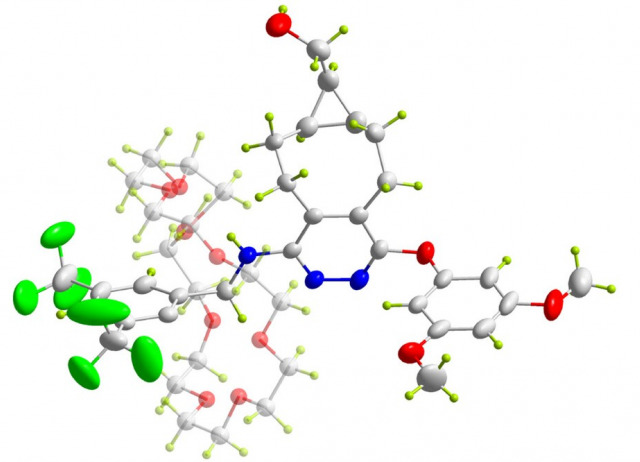
(S)-BINOL-derived chiral picolinate ligand as a platform for new polymer frameworks: a pilot study on the effect of the binaphthyl scaffold vs picolinate
This work proposes a graphical analysis based on a new chiral picolinate of the (S)-BINOL derivative, designated as the reference ligand L1. The models establish the influence of temperature on L1 and identify a new coordination polymer (CP) [Cu2(pic)2(OTf)2(H2O)2(Py)]n_2n(Py) 1.2(Py), where [Hpic = pyridine 2-carboxylic acid and pic = picolinate], as the predominant structure through the pathway in dioxane at room temperature or the known complex [Cu(pic)2.2H2O] 2.(H2O)2 at reflux in dioxane. This work provides a detailed description of the preparation method, the structural characteristics of the new CP containing the zinc complex [Zn2(pic-Hpic)3]n.n(OTf) 3.(OTf), and density functional theory (DFT) calculations. Coordination polymer 1 2(Py) exhibits unusual magnetic properties at low temperature and high magnetic fields. This behavior may be associated with the presence of superparamagnetic particles or the influence of crystal field effects. In contrast, its origin is unlikely to be associated with common ferromagnetic impurities such as Fe, Co, or Ni, given its characteristic high Curie temperature.
Humberto A. Rodríguez, Daniel A. Cruz, Juan I. Padrón and Pablo Lorenzo-Luis
Alginate Microencapsulation as a Tool to Improve Biostimulant Activity Against Water Deficits
Climate change is reducing agricultural productivity through altered weather patterns and extreme events, potentially decreasing yields by 10–25%. Biostimulants like pyroglutamic acid can enhance plant tolerance to water stress, but their rapid degradation in the soil limits effectiveness. Encapsulation in alginate matrices promises to be a good solution, protecting the compound and enabling controlled release. This study reports, for the first time, that encapsulated pyroglutamic acid markedly enhances drought tolerance in tomato and maize plants. The encapsulation strategy reduces effective concentration by an order of magnitude while significantly improving water use efficiency, photo-synthetic performance, and overall stress resilience. These findings demonstrate that alginate-based encapsulation substantially increases biostimulant uptake and efficacy, providing a novel and efficient strategy to mitigate water stress in crops, with important implications for climate-resilient agriculture. Two encapsulation methods for generating the alginate microcapsules are compared: ionic gelation with Nisco® system and the electrospray technique.
Jiménez-Arias, David; Morales-Sierra, Sarai; García-García, Ana L.; Herrera, Antonio J.; Pérez Schmeller, Rayco; Suárez, Emma; Santana-Mayor, Álvaro; Silva, Patrícia; Borges, João Paulo; Pinheiro de Carvalho, Miguel Â. A.
Cysteine Alkylation in Enzymes and Transcription Factors: A Therapeutic Strategy for Cancer
Metabolic enzymes and cancer-driving transcriptions factors are often overexpressed in neoplastic cells, and their exposed cysteine residues are amenable to chemical modification. This review explores cysteine alkylation as a cancer treatment strategy, focusing on Michael acceptors like curcumin and helenalin, which interact with transcription factors NF-κB, STAT3 and HIF-1α. Molecular docking studies using AutoDockFR revealed distinct binding affinities: curcumin showed strong interactions with STAT3 and NF-κB, while helenalin exhibited high affinity for STAT3 and HIF-1α. Synthetic compounds like STAT3-IN-1 and CDDO-Me demonstrated superior binding in most targets, except for CDDO-Me with HIF-1α, suggesting unique structural incompatibilities. Natural products such as zerumbone and umbelliferone displayed moderate activity, while palbociclib highlighted synthetic-drug advantages. These results underscore the importance of ligand−receptor structural complementarity, particularly for HIF-1α’s confined binding site, where helenalin’s terminal Michael acceptor system proved optimal. The findings advocate for integrating computational and experimental approaches to develop cysteine-targeted therapies, balancing synthetic precision with natural product versatility for context-dependent cancer treatment strategies.
Curieses Andrés, Celia María; Lobo, Fernando; Pérez de Lastra, José Manuel; Bustamante Munguira, Elena; Andrés Juan, Celia; Pérez-Lebeña, Eduardo.
Pesticide Residues and Mycotoxins in Natural and Conventional Wines: Insights from Italy and Spain
Natural wine is a captivating and rapidly evolving phenomenon in the world of viticulture and winemaking. This movement, characterized by minimal intervention, low or no chemical additives, and a focus on terroir-driven expressions of grapes, has ignited a passionate following among wine enthusiasts, sommeliers, and eco-conscious consumers. It's more than just a beverage; it embodies a philosophy that aligns with contemporary sustainability and wellness trends. Though natural wine enthusiasts have produced ample coverage of the natural wine movement in books, essays and films, academics have lagged behind in their examination of this global phenomenon. In Uncorked: Negotiating Science and Belief in the Natural Wine Movement, scholarly discussion reaches across disciplines to offer valuable insights for researchers, producers, wine enthusiasts and anyone with an interest in this movement. Exploring the relevance of natural wine offers a unique lens through which to examine the intersection of tradition, innovation, environmental stewardship, and the evolving tastes of modern consumers. Through the stories of the winemakers, the science behind natural fermentation, and the cultural and historical context, this book aims to provide a comprehensive look at how this movement is shaping the future of wine and our relationship with the natural world. Each essay from a thought leader in their respective field delves deep into the historical, ecological, and sociocultural dimensions of natural winemaking, shedding light on its ancient roots, ecological practices, and the philosophical underpinnings that drive the movement. Discussions of regulatory and consumer aspects will also elucidate the practical considerations of making and selling natural wine. Uncorked: Negotiating Science and Belief in the Natural Wine Movement is a groundbreaking edited book that offers a comprehensive and scholarly exploration ofthe world of natural wine. This interdisciplinary volume brings together leading experts in fields ranging from enology and sensory studies to anthropology and philosophy, creating a rich tapestry of insights into the production, consumption, and cultural significance of natural wine. Academics, winemakers and wine enthusiasts alike will be illuminated by this volume.
Alonso-González, Pablo; Acosta-Dacal, Andrea; Zaccaroni, Annalisa; Luzardo, Octavio P.
Interview with Aaron Ayscough
Natural wine is a captivating and rapidly evolving phenomenon in the world of viticulture and winemaking. This movement, characterized by minimal intervention, low or no chemical additives, and a focus on terroir-driven expressions of grapes, has ignited a passionate following among wine enthusiasts, sommeliers, and eco-conscious consumers. It's more than just a beverage; it embodies a philosophy that aligns with contemporary sustainability and wellness trends. Though natural wine enthusiasts have produced ample coverage of the natural wine movement in books, essays and films, academics have lagged behind in their examination of this global phenomenon. In Uncorked: Negotiating Science and Belief in the Natural Wine Movement, scholarly discussion reaches across disciplines to offer valuable insights for researchers, producers, wine enthusiasts and anyone with an interest in this movement. Exploring the relevance of natural wine offers a unique lens through which to examine the intersection of tradition, innovation, environmental stewardship, and the evolving tastes of modern consumers. Through the stories of the winemakers, the science behind natural fermentation, and the cultural and historical context, this book aims to provide a comprehensive look at how this movement is shaping the future of wine and our relationship with the natural world. Each essay from a thought leader in their respective field delves deep into the historical, ecological, and sociocultural dimensions of natural winemaking, shedding light on its ancient roots, ecological practices, and the philosophical underpinnings that drive the movement. Discussions of regulatory and consumer aspects will also elucidate the practical considerations of making and selling natural wine. Uncorked: Negotiating Science and Belief in the Natural Wine Movement is a groundbreaking edited book that offers a comprehensive and scholarly exploration ofthe world of natural wine. This interdisciplinary volume brings together leading experts in fields ranging from enology and sensory studies to anthropology and philosophy, creating a rich tapestry of insights into the production, consumption, and cultural significance of natural wine. Academics, winemakers and wine enthusiasts alike will be illuminated by this volume.
Ayscough, Aaron; Alonso-González, Pablo.